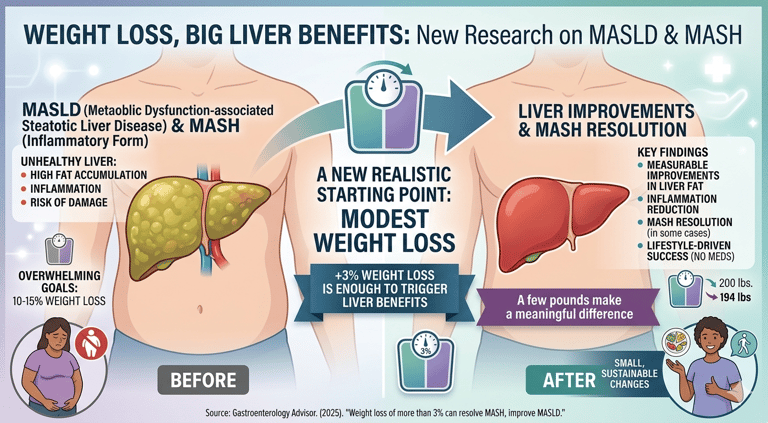
Key Findings
Weight loss of more than 3% was linked to measurable improvements in liver fat and inflammation. Patients who achieved this modest reduction were significantly more likely to see MASH resolve compared with those who didn’t lose weight. Greater weight loss produced even stronger benefits, but the study emphasizes that small, achievable changes still matter. Improvements were observed even without medications, showing the powerful impact of lifestyle‑driven weight reduction.
Why This Matters
MASLD and MASH are increasingly common, often tied to:
metabolic syndrome
insulin resistance
obesity
sedentary lifestyle
Many patients feel overwhelmed when told they need to lose 10–15% of their body weight to reverse liver disease. This study offers a more realistic starting point: even a few pounds can make a difference.
What the Research Suggests
Weight loss doesn’t need to be extreme to be effective.
Early improvements in liver health can occur with small, consistent changes.
This may help motivate patients who struggle with long‑term weight‑loss goals.
The Bigger Picture
While medications for MASH are emerging, lifestyle changes remain the foundation of treatment. This study reinforces that modest, sustainable weight loss is both achievable and clinically meaningful.
A new analysis highlights something surprisingly encouraging: you don’t need dramatic weight loss to improve metabolic dysfunction–associated steatotic liver disease (MASLD).
In fact, losing just over 3% of body weight may be enough to trigger meaningful improvements — and in some cases, even resolve MASH, the inflammatory form of the disease.
Weight Loss, Big Liver Benefits — New Research on MASLD & MASH
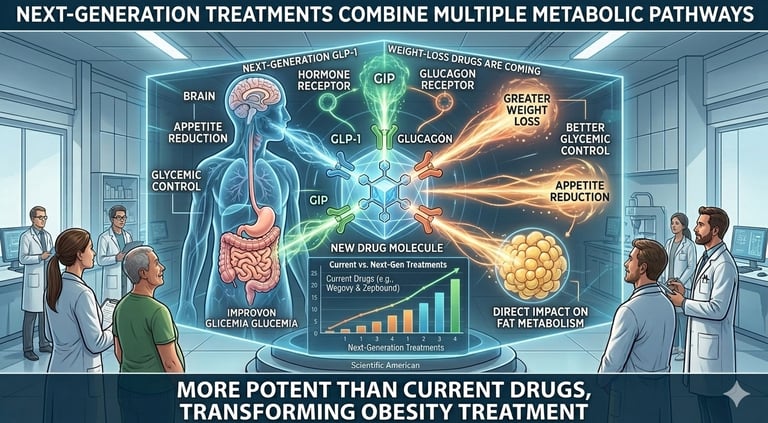
These new drugs aim to:
produce greater weight loss than current GLP‑1s
improve blood sugar control
reduce appetite more effectively
target fat metabolism more directly
Some of the new medications in development activate two or even three hormone receptors at once, such as GLP‑1, GIP, and glucagon receptors. Early clinical trials suggest these combinations may lead to faster and deeper weight‑loss effects than existing drugs.
Experts believe these next‑generation therapies could reshape obesity treatment by offering stronger results, fewer side effects, and more personalized options.
Scientific American. (2025). New GLP‑1 weight‑loss drugs are coming—and they’re stronger than Wegovy, Zepbound.
New GLP‑1 weight‑loss drugs are on the way — and they may be even more powerful than today’s blockbuster medications like Wegovy and Zepbound.
Researchers and pharmaceutical companies are developing next‑generation treatments that combine multiple metabolic pathways in a single medication.
New GLP‑1 weight‑loss drugs are on the way
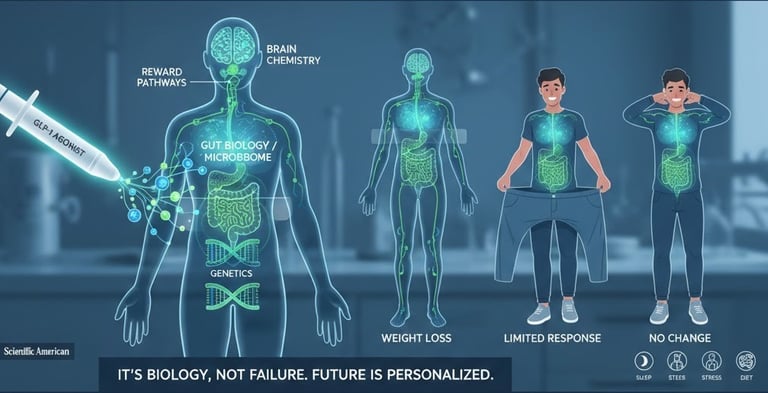
Researchers have found that:
Some people have genetic variations that make GLP‑1 signaling less effective.
Differences in the gut microbiome may influence how strongly the drugs suppress appetite.
People with long‑term obesity may have brain‑reward pathways that are harder to shift.
Lifestyle factors — sleep, stress, diet — can change how well the medication works.
The big takeaway: If someone doesn’t lose weight on a GLP‑1 drug, it’s not a personal failure — it’s biology. Future treatments may be more personalized, targeting multiple hormones or metabolic pathways to help more people respond.
Scientific American. (2025). Why Ozempic and Wegovy don’t cause weight loss for everyone
Not everyone loses weight on Ozempic or Wegovy — and scientists are finally uncovering why. These medications work by mimicking GLP‑1, a hormone that reduces appetite and helps regulate blood sugar. But people respond very differently because of genetics, gut biology, metabolism, and brain chemistry.
Why Ozempic and Wegovy don't cause weight loss in everyone.
Welfare
Our holistic perspective ensures that we take into account both physical and emotional well-being, allowing us to create personalized treatment plans and supportive environments for our clientele.
Health
balance
© 2025. All rights reserved.
1077 Celestial St Suite 204 Cincinnati OH
Phone (513) 346-8455
